|
POLYMER HISTORY |
| As far back as 1839, Charles Goodyear first improved the elastic
properties of natural rubber by heating with sulfur (vulcanization). It was not
until the 1930s that the macromolecule model of rubber was understood. After
World War II and through the 1950s rapid developments in synthetic polymers
were made. Most commercial high-performance elastomers trace their origins to
the 1960s and 1970s.
Polymers are long chains of repeating chemical units, or monomers. The chemical skeletal structures may be linear, cyclic or branched. When one monomer is polymerized, the resultant polymer is called a homopolymer. Examples include polyethylene, polystyrene and polytetrafluoroethylene (PTFE). Copolymers (or dipolymers) are derived from the polymerization of more than one type of monomer. The distribution of monomers in these copolymers can be statistical, random or alternating. Examples include ethylene - propylene and fluorocarbon elastomers (vinylidene fluoride and hexafluoropropylene). Terpolymers are three - monomer - unit polymers, such as ethylene - propylene - diene (EPDM) and specialty fluorocarbon grades. |
TYPES OF POLYMERS |
| There are three general classes of
polymers:
1. Thermoplastics (can be melted with the application of heat)
2. Thermosets (degrade rather than melt with the application of heat) 3. Elastomers (cross-linked)
Plastics are rigid long-chain polymers which are
not usually
Thermoplastic elastomers (TPEs) often combine
the properties of elastomers with |
RUBBER PROCESSING |
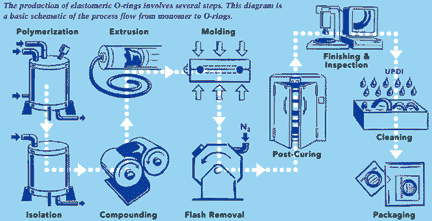
| Polymerization |
The beginning step for elastomers is the
polymerization of the backbone and cure-site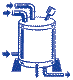 monomers. This is typically done by large
chemical companies such as Du Pont, Dow,
GE, Ausimont, Daikin and Dyneon. Common
techniques are emulsion, microemulsion, and
suspension polymerization. Polymerization
combines two or more process gases (monomers) into an aqueous
environment and under specific temperature and pressure
conditions connects the individual monomers into the desired
polymer. Initiating agents, buffers and other chemicals may be
added to the polymer reactor to achieve the desired chemical
properties and polymerization dynamics.
monomers. This is typically done by large
chemical companies such as Du Pont, Dow,
GE, Ausimont, Daikin and Dyneon. Common
techniques are emulsion, microemulsion, and
suspension polymerization. Polymerization
combines two or more process gases (monomers) into an aqueous
environment and under specific temperature and pressure
conditions connects the individual monomers into the desired
polymer. Initiating agents, buffers and other chemicals may be
added to the polymer reactor to achieve the desired chemical
properties and polymerization dynamics. |
| Isolation | |
The backbone polymers are isolated (brought out
of the emulsion), cleaned and dried.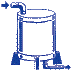 Chemical
agents may be added at this step to isolate the
polymer �latex� into a more usable form.
Once the polymer is cleaned and dried, the
�crumb� polymer is shipped to compounders
(or O-ring molders) for mixing. Chemical
agents may be added at this step to isolate the
polymer �latex� into a more usable form.
Once the polymer is cleaned and dried, the
�crumb� polymer is shipped to compounders
(or O-ring molders) for mixing. |
|
| Compounding (mixing) |
The �crumb� polymer is mixed with a cross-linking agent and
other functional fillers. The cross-linking agent allows  chemical
bonds to form between the polymer backbones,
thus providing resiliency to the material.
Functional fillers include reinforcing fillers, pigments, anti-degradants, acid scavengers and
process aids. These ingredients are typically
mixed together on a 2-roll mill or other custom
mixing machinery. chemical
bonds to form between the polymer backbones,
thus providing resiliency to the material.
Functional fillers include reinforcing fillers, pigments, anti-degradants, acid scavengers and
process aids. These ingredients are typically
mixed together on a 2-roll mill or other custom
mixing machinery. |
| Types of Polymerization
Reactions
1. Condensation Polymerization� yields polymers with repeating units having fewer atoms than the monomers from which they are formed. This reaction generally involves the elimination of small molecules such as H2O or HCl. 2. Addition Polymerization 3. Chain Polymerization/Free Radical Polymerization�
Types of Chain Polymerization Methods
|
| Once the material is compounded,
it is shaped into sheets and then
shipped to O-ring molders:
|
 Curing
Some high-performance elastomers are subjected to a post-curing
operation. Elastomer parts are exposed to high
temperatures in carefully controlled environments
for several hours to complete the curing process.
Additionally, this post-curing step removes excess
water vapor and volatile process additives, thereby
improving vacuum and contamination performance. Curing
Some high-performance elastomers are subjected to a post-curing
operation. Elastomer parts are exposed to high
temperatures in carefully controlled environments
for several hours to complete the curing process.
Additionally, this post-curing step removes excess
water vapor and volatile process additives, thereby
improving vacuum and contamination performance.
|
COMMON POLYMERS |
| ASTM | Polymer | Trade Names | Monomers |
|
PLASTICS |
|||
| Polyamideimide (PAI) | TORLON� | ||
| Polybenzimidazole (PBI) | CELAZOLE� | -(C7H6N2)- | |
| Polycarbonate (PC) | -COOC6H5C(CH3)2C6H5O- | ||
| Polyethylene (PE) | -CH2CH2- | ||
| Polyetheretherketone (PEEK) |
KETRON� |
-C6H5-CO-C6H5-O-C6H5- | |
| Polyetrherimide (PEI) |
ULTEM� |
||
| Polyimide (PI) |
DURATRON� |
N(C2O2)C6H5(C2O2)N-R- | |
| Polypropylene (PP) | CH2CH(CH3)- | ||
| Polyphenylenesulfide (PPS) |
TECHTRON� |
||
| Polyvinylidine Fluoride (PVDF) | -CH2CF2- | ||
| Fluorinated Ethylene-Propylene (FEP) |
TEFLON� FEP |
-CF2CF2-CF2CF(CF3)- | |
| Perfluoroalkoxy (PFA) |
TEFLON� PFA |
-CF2CF2-CF2CF(OCF3)- | |
| Polytetrafluoroethylene (PTFE) |
TEFLON� PTFE |
-CF2CF2- | |
|
ELASTOMERS |
|||
| NBR | Nitrile (Buna-N) | PARACRIL�, CHEMIGUM� | -CH2CH=CH(CH2)2CHCH(CN)- |
| EPDM | Ethylene-Propylene Diene | VISTALON�, NORDEL� | -CH2CH2-CH2CH(CH3)- |
| VMQ | Silicone | SILASTIC�, SILPLUS� | -OSi(CH3)2-OSi(CH3)(CH=CH2)- |
| FVMQ | Fluorosilicone | SILASTIC� LS, FSE� | -OSi(CH3)(CH=CH2)-OSi(CH3) (CH2CH2CF3)- |
| FKM | Fluoroelastomer A | VITON�, FLUOREL� | -CH2CF2-CF2CF(CF3)- |
| Fluoroelastomer B | -CH2CF2-CF2CF(CF3)-CF2CF2- | ||
| Fluoroelastomer GF | VITON�, ETP� | -CF2CF2-CF2CF(OCF3)-CH2CH2- | |
| Fluoroelastomer TFE/P | AFLAS� | -CF2CF2-CH2CH(CH3)- | |
|
FFKM |
Perfluoroelastomer | AEGIS�, CHEMRAZ�, KALREZ� | -CF2CF2-CF2CF(OCFnCF3)- |
|
OTHER |
|||
| VF2/CTFE |
KEL-F� |
-CH2CF2-CF2CFCI- | |
SEALING ELASTOMERS |
| Sealing elastomers may best be initially understood and compared by
examining their chemical structure. It is this chemical structure that forms
the foundation for a seal�s ability to withstand certain chemical, thermal or
physical environments.
The seal industry uses many tests to determine an elastomer�s chemical and thermal compatibility as well as physical properties which can have a great influence on the performance in high-pressure or vacuum environments. These properties can provide an insight into the mode of degradation or the retention of sealing properties�all useful information in predicting seal life or comparing economic alternatives. Another difference in elastomer compounds is the compounding (or mixing) of ingredients. These factors can provide unique pigmentation, improved specific chemical or thermal properties, improved dynamic performance, reduced cost, improved electrical properties, reduced friction or sticking, and many other aspects of seal performance. |
 connected or cross-linked. Plastics
can either be thermoplastic�meaning they
can be heated and cooled without changing
properties�or thermoset, where an increase in
temperature changes the chemical structure and properties. As a
class, plastics have low elongation and high elongation set.
connected or cross-linked. Plastics
can either be thermoplastic�meaning they
can be heated and cooled without changing
properties�or thermoset, where an increase in
temperature changes the chemical structure and properties. As a
class, plastics have low elongation and high elongation set. Elastomers
are flexiblelong - chain polymers
which are capable of cross-linking. Cross-linking
chemically bonds polymer chains which can
prevent reversion to a non-cross-linked
polymer at elevated temperatures. The cross-link is the key to
the elastic, or rubbery, properties of these materials. The elasticity
provides resiliency in sealing applications.
Elastomers
are flexiblelong - chain polymers
which are capable of cross-linking. Cross-linking
chemically bonds polymer chains which can
prevent reversion to a non-cross-linked
polymer at elevated temperatures. The cross-link is the key to
the elastic, or rubbery, properties of these materials. The elasticity
provides resiliency in sealing applications. the ease of
processability of thermoplastics. They are the result
of a physical combination of soft, elastic polymer
segments and hard, crystalline
segments which are
capable of cross-linking. Thermoplastic elastomers are generally
classified by their structure rather than their chemical makeup.
the ease of
processability of thermoplastics. They are the result
of a physical combination of soft, elastic polymer
segments and hard, crystalline
segments which are
capable of cross-linking. Thermoplastic elastomers are generally
classified by their structure rather than their chemical makeup. Extrusion
The sheet compound
is extruded into
a configuration
similar to the desired
finished part.
Extrusion
The sheet compound
is extruded into
a configuration
similar to the desired
finished part.  Molding
Most of the elastomeric O-rings used in
the semiconductor industry are compres-sion
molded. A preshaped form is inserted
into a multi-section
mold and trans-ferred
to a heated
press. Under heat
and pressure, the
elastomer flows into the mold cavities and chemical cross-linking takes place
(or begins to take place, depending on the specific elastomer
compound). After a period of time ranging from several seconds
to several minutes, the parts are removed from the hot molds.
Depending on the compound, mold releases are often used. These
diluted spray coatings are often a derivative of fluoropolymers, or
silicone-based polymers.
Molding
Most of the elastomeric O-rings used in
the semiconductor industry are compres-sion
molded. A preshaped form is inserted
into a multi-section
mold and trans-ferred
to a heated
press. Under heat
and pressure, the
elastomer flows into the mold cavities and chemical cross-linking takes place
(or begins to take place, depending on the specific elastomer
compound). After a period of time ranging from several seconds
to several minutes, the parts are removed from the hot molds.
Depending on the compound, mold releases are often used. These
diluted spray coatings are often a derivative of fluoropolymers, or
silicone-based polymers.  Flash Removal
After the parts are removed from the molds,
they contain thin �flash� as a result of the
elastomer flowing in the multi-section mold.
This �flash� is typically removed by exposing
the parts to a cryogenic tumbling process. The
elastomer is cooled and tumbled, causing the
thinner �flash� section to become brittle and
break away from the main part. Additional tumbling or hand-deflashing
may be required on some part designs or compounds.
Flash Removal
After the parts are removed from the molds,
they contain thin �flash� as a result of the
elastomer flowing in the multi-section mold.
This �flash� is typically removed by exposing
the parts to a cryogenic tumbling process. The
elastomer is cooled and tumbled, causing the
thinner �flash� section to become brittle and
break away from the main part. Additional tumbling or hand-deflashing
may be required on some part designs or compounds. Finishing and Inspection
After the parts are removed from the curing
ovens, the parts are again cleaned and
inspected to ensure the parts meet the
material and dimensional specifications.
Finishing and Inspection
After the parts are removed from the curing
ovens, the parts are again cleaned and
inspected to ensure the parts meet the
material and dimensional specifications.  Cleaning
After the parts are inspected, acceptable parts are
delivered to the Class 100 clean room for cleaning
and packaging. An ultrapure
deionized water (UPDI)
rinsing cycle removes
surface contamination
from the parts.
Cleaning
After the parts are inspected, acceptable parts are
delivered to the Class 100 clean room for cleaning
and packaging. An ultrapure
deionized water (UPDI)
rinsing cycle removes
surface contamination
from the parts.  Packaging
Acceptable parts are then
counted and packaged, either
individually or in bulk, in a
heat-sealed clean inner bag.
The parts are then packaged in
an outer bag, with a complete
description of the parts, lot
number, the batch and cure date, as well as any
specific information relative to the customer�s
part number.
Packaging
Acceptable parts are then
counted and packaged, either
individually or in bulk, in a
heat-sealed clean inner bag.
The parts are then packaged in
an outer bag, with a complete
description of the parts, lot
number, the batch and cure date, as well as any
specific information relative to the customer�s
part number.